Prototype of running clinical trials in an untrustworthy environment using blockchain | Nature Communications

Sharing and reuse of individual participant data from clinical trials: principles and recommendations | BMJ Open

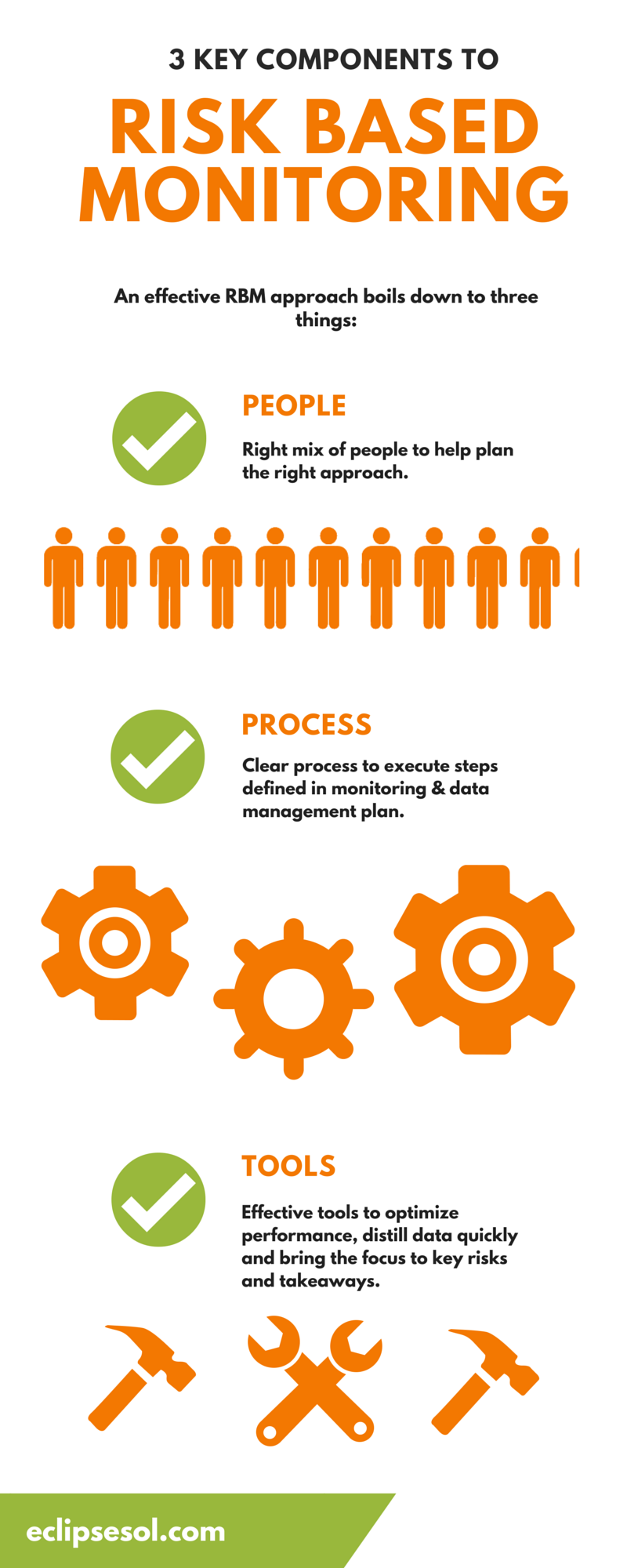
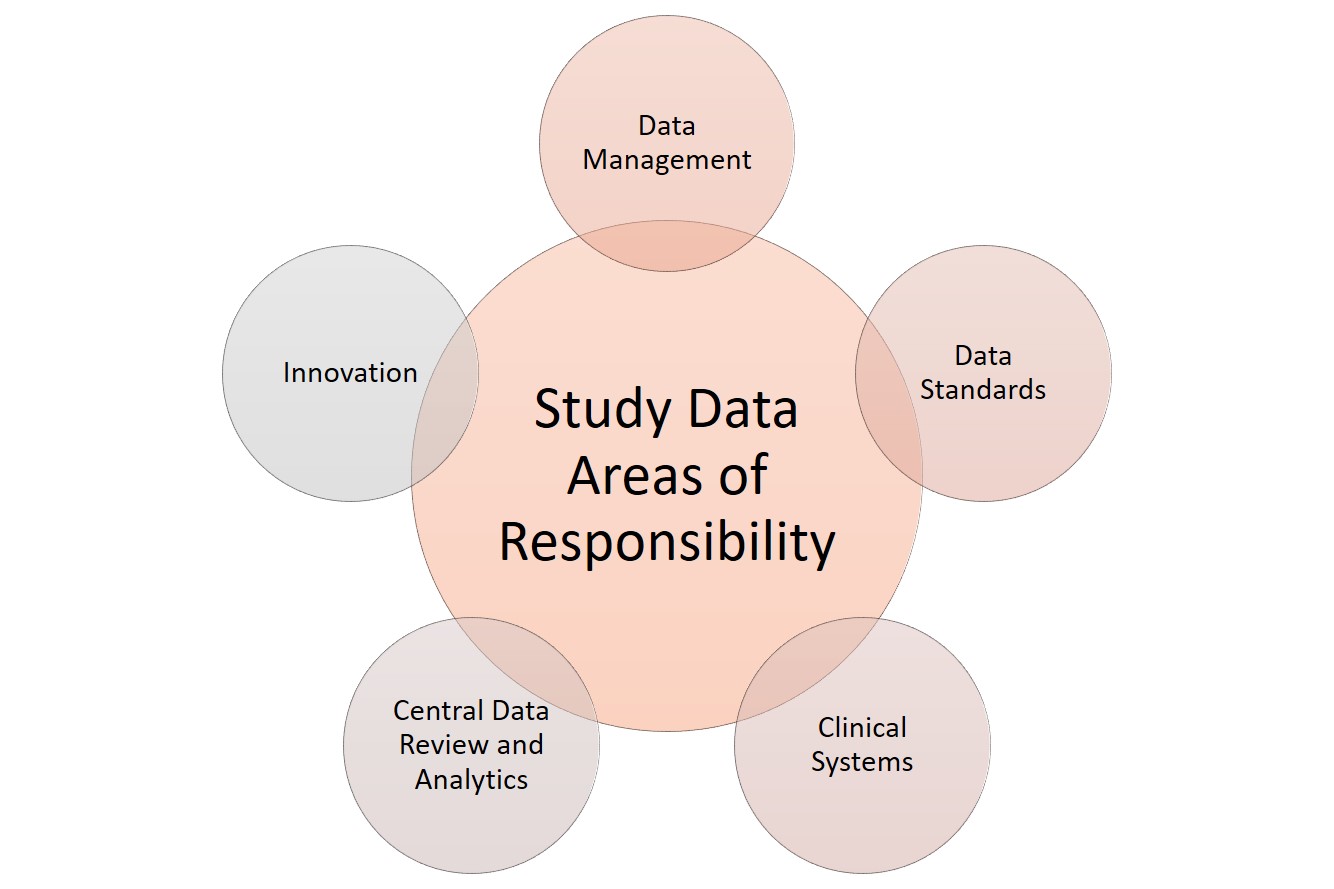
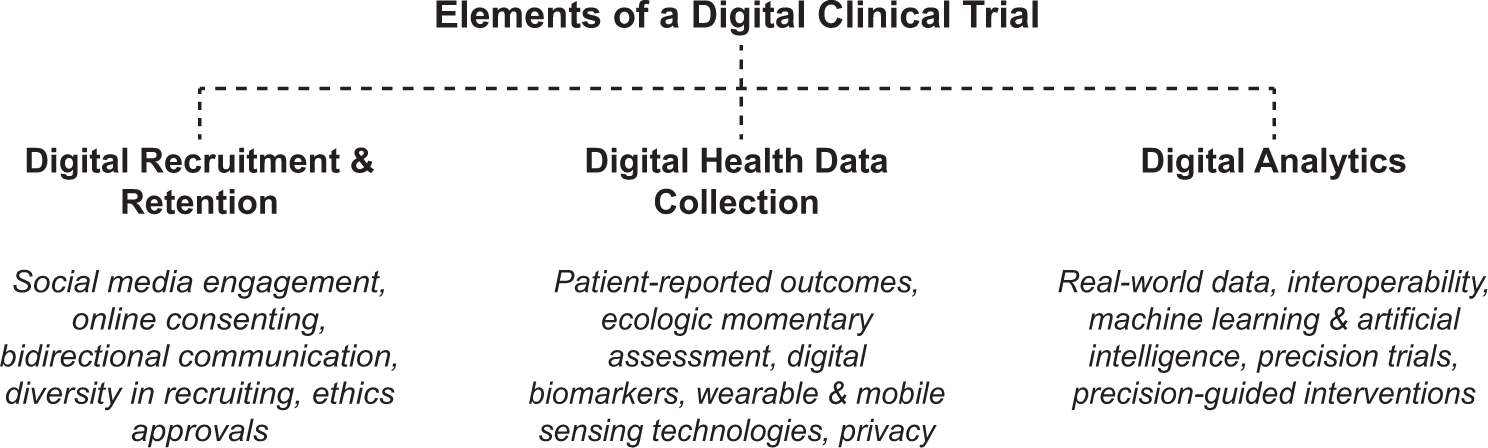
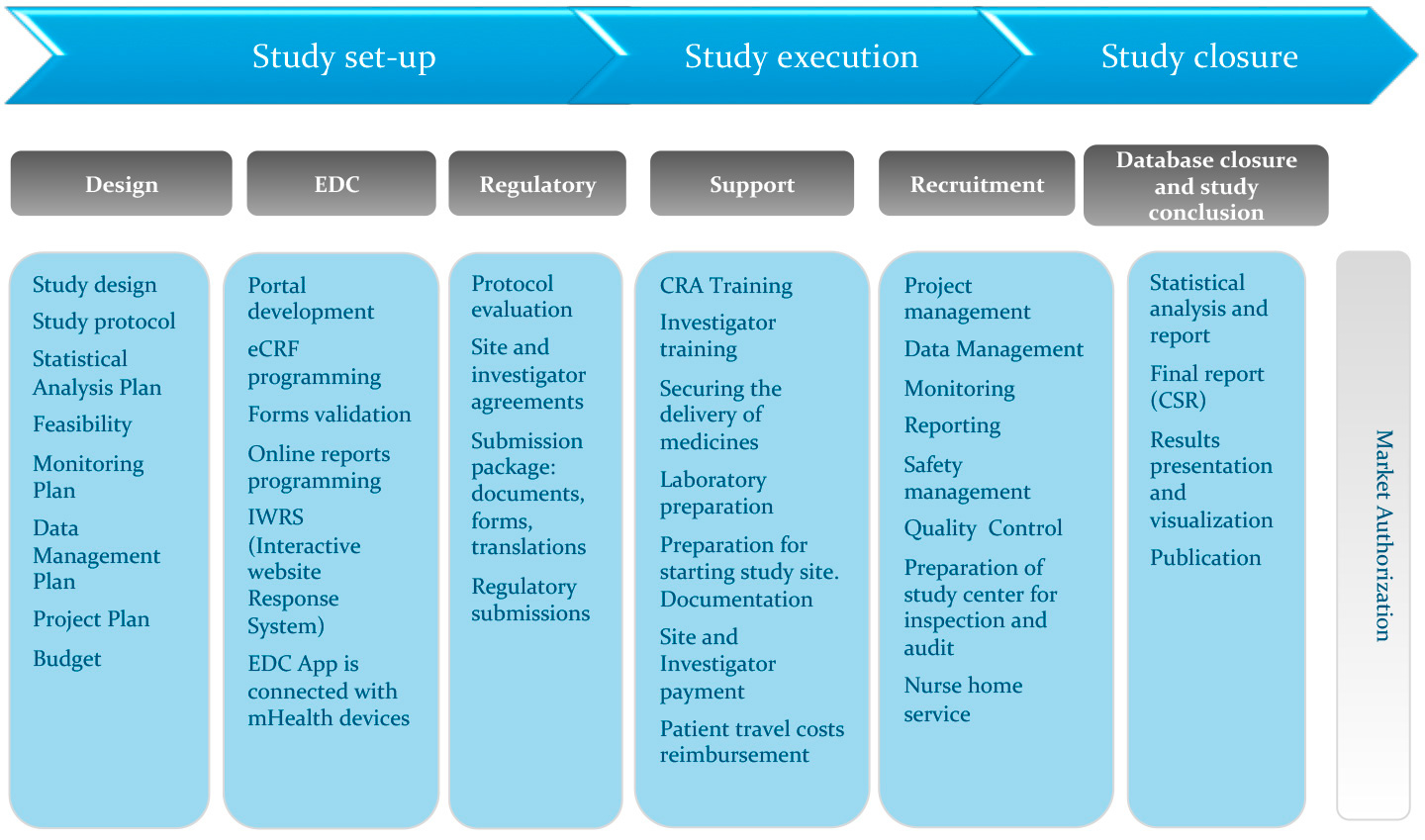
Safety Management Plan – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India